FDA 510(k) submissions and EU MDR conformity assessments are generally built around the complete medical device and its intended use. At the same time, how a product is classified depends on how it is defined, labeled, and brought to market. Understanding that difference helps explain why components like medical foot switches are typically not submitted independently, and what role they play in supporting the overall regulatory process.
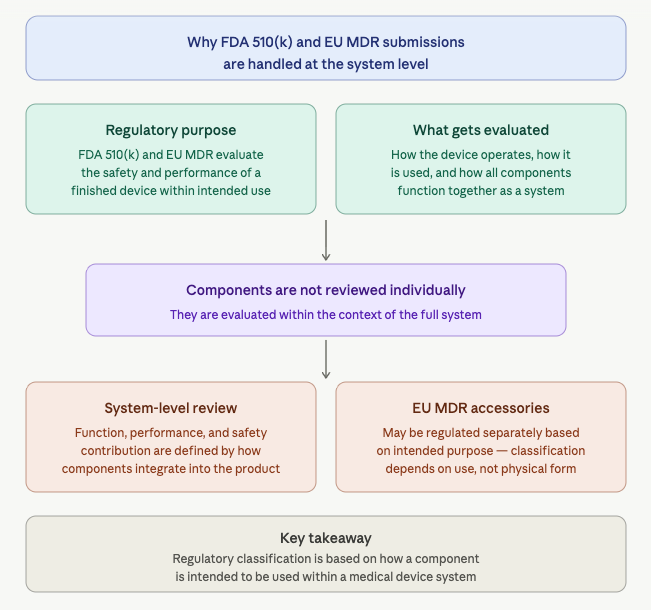
Why FDA 510(k) and EU MDR Submissions Are Handled at the System Level
Regulatory pathways such as FDA 510(k) and EU MDR are designed to evaluate the safety and performance of a finished medical device within its intended use.
That evaluation looks at how the device operates, how it is used, and how all components function together as a complete system. Because of this, many medical device components are not reviewed on their own when they are being supplied for integration into another manufacturer’s device.
Instead, they are evaluated within the context of the full system. Their function, performance, and contribution to safety are defined by how they are integrated into the final product.
Under EU MDR, the framework also includes medical device accessories, which may be regulated separately depending on their intended purpose and how they are brought to market. This means classification is not based only on the physical component, but on how it is intended to be used within a medical device system.

Why Linemaster Does Not Submit a 510(k)
Foot switches supplied by Linemaster Switch Corporation are typically provided as components for integration into a customer’s finished medical device, rather than being marketed as standalone medical devices or finished medical device accessories.
The same medical foot switch can be used across multiple applications, each with a different intended use, operating environment, and regulatory classification. Because of that, submitting a foot switch on its own would not reflect how it is actually used in practice.
A 510(k) submission requires a clearly defined device with a specific intended use and full system context. That level of definition is established by the finished device manufacturer, not at the individual component level.
For that reason, responsibility for FDA 510(k) submission or EU MDR conformity assessment remains with the device manufacturer, who defines how the full system functions and how each component is applied.
How Linemaster Supports Customer Submissions
While Linemaster does not typically submit a 510(k) for individual components, the medical foot switches provided still play a direct role in supporting customer regulatory submissions.
Support starts with delivering consistent, well-defined components that can be reliably integrated into a customer’s device. Keeping alignment between design specifications and production ensures that what is validated during development matches what is ultimately used in the final device and included in regulatory submissions.
Linemaster also performs internal testing to understand performance, durability, and expected use conditions. This testing provides objective data that supports system-level verification and validation activities carried out by the medical device manufacturer.
In addition, technical documentation is provided that can be included in a customer’s design history file or EU MDR technical documentation. This may include product specifications, performance data, and traceability information that help define how the component behaves within the overall system.
By focusing on consistency, documentation, and repeatable performance, Linemaster supports customers in integrating medical foot switches into their devices with a clear understanding of regulatory expectations and system performance.

How Customers Typically Approach FDA 510(k) and EU MDR Submissions
For medical device manufacturers, the regulatory submission process is centered around the complete system.
This includes defining the intended use, validating overall performance, and demonstrating that the finished medical device meets applicable regulatory requirements. Components like foot switches are included as part of that system and evaluated within that context.
In some cases, depending on intended use and how a product is brought to market, a component may instead be classified as a medical device accessory and subject to its own regulatory requirements. That determination is based on regulatory definitions and labeling, not just the form of the component itself.
Why This Distinction Matters in Medical Device Design
Understanding how FDA 510(k) and EU MDR submissions are structured helps reduce confusion during product selection and system design.
Instead of focusing on whether a component has its own regulatory approval, the more important consideration is whether it is suitable for integration into a regulated medical device. This comes down to consistency, documentation, and predictable performance.
Selecting well-characterized components supports a smoother development process and helps ensure that the final submission reflects how the device will actually be used.
Conclusion
FDA 510(k) submissions and EU MDR conformity assessments are generally built around complete medical devices, not individual components.
Medical foot switches are typically evaluated as part of the systems they support when they are integrated into a finished device. At the same time, regulatory treatment depends on intended purpose and how the product is brought to market, especially under EU MDR where accessories may be regulated separately.
Linemaster Switch Corporation supports this process by providing consistent, well-documented components and the technical information needed for successful integration into customer devices. This approach allows manufacturers to include medical foot switches in their submissions with confidence in performance, reliability, and overall system alignment.
Meet The Author

Arijan Kandic
Digital Marketing Specialist
Arijan is the Digital Marketing Specialist at Linemaster Switch Corporation and holds a bachelor’s degree in business management from Quinnipiac University. He manages the company’s SEO strategy, Google Ads campaigns, and digital marketing initiatives, and develops educational content for the Linemaster Learning Center to help engineers, OEMs, and medical device manufacturers better understand foot switch technology. Arijan works closely with Linemaster’s engineering and applications teams to translate complex technical concepts into clear, accurate articles on foot switch design, customization, and compliance considerations.
In Collaboration with

Sean Lewis
Director of Engineering
Sean has more than fifteen years of experience in product development, engineering governance, and cross functional technical operations. His background in metal fabrication, including machining, forming, welding, and inspection, provides a strong manufacturing foundation that supports his approach to design and process optimization. Sean holds a bachelor’s degree in mechanical engineering, an MBA with a manufacturing concentration, and an MSOL. He is a Certified SolidWorks Expert with advanced capability in CAD, rendering, simulation, and rapid prototyping. Sean also specializes in DFMEA and PFMEA risk management practices and is the holder of several foot switch design and utility patents.
Uploaded 05/11/2026
Custom Foot Switches
Linemaster’s custom footswitches are designed to meet specific user requirements, offering a range of features such as various pedal configurations, wired and wireless options, and customizable LED indicators. These custom footswitches provide reliable, durable solutions tailored to enhance functionality in diverse applications.


